Eli Lilly Stock Analysis: Explosive Growth, R&D Pipeline Momentum, and Increased 2025 guidance
Eli Lilly and Company (NYSE: LLY) delivered another impressive quarter, exceeding expectations and demonstrating the unstoppable momentum of its incretin portfolio. The company's Q3 2025 financial results, released on October 30, 2025, highlighted massive revenue growth, a strong pipeline, and a substantial boost to full-year guidance.
Q3 Financial Overview
Lilly’s financial performance in Q3 2025 was nothing short of exceptional. Worldwide revenue surged an astounding 54% year-over-year to $17.60 billion, primarily driven by massive volume growth. Key Products revenue grew to $11.98 billion in Q3 2025, led by Mounjaro and Zepbound.
-
Mounjaro: worldwide revenue rocketed by 109% to $6.52 billion. U.S. revenue was $3.55 billion, an increase of 49%, reflecting strong demand.
-
Zepbound: U.S. revenue saw a staggering 184% increase to $3.57 billion, compared with $1.26 billion in Q3 2024.
This top-line strength cascaded down to profitability, with non-GAAP Earnings Per Share (EPS) climbing by $5.84 to $7.02, compared with EPS of $1.18 in Q3 2024. Non-GAAP EPS in Q3 2025 and Q3 2024 included acquired IPR&D charges of $0.71 and $3.08, respectively.
In response to this sustained momentum, Lilly increased its 2025 full-year revenue guidance to a range of $63.0 billion to $63.5 billion, compared to the prior's $60.0 billion to $62.0 billion, and raised its non-GAAP EPS guidance to between $23.00 and $23.70, compared to the prior's $21.75 to $23.00. This is a powerful signal of management’s confidence in its commercial execution and capacity expansion.
Pipeline and Operational Strength
Beyond the financials, Lilly made crucial progress in both its pipeline and manufacturing capabilities, addressing the market's key concerns: supply and future growth.
-
Orforglipron Advancement: The oral GLP-1 asset achieved positive results across four Phase 3 trials in type 2 diabetes and obesity. Critically, Lilly announced plans to submit orforglipron for global regulatory approval for obesity by the end of the year.
-
Regulatory Progress: The U.S. FDA approval of Inluriyo (imlunestrant) for certain advanced or metastatic breast cancer adds another critical revenue driver to the portfolio.
-
Capacity Expansion: Lilly announced plans for two new manufacturing facilities in Virginia and Texas, along with an expansion of its existing Puerto Rico site. This multi-billion-dollar investment signals a strong commitment to meeting the insatiable global demand for its incretin drugs.
In addition to pipeline successes across obesity, diabetes, oncology, and Alzheimer’s, Lilly remains aggressive in innovation partnerships, including an AI-driven drug discovery collaboration with NVIDIA and continued biotech investments.
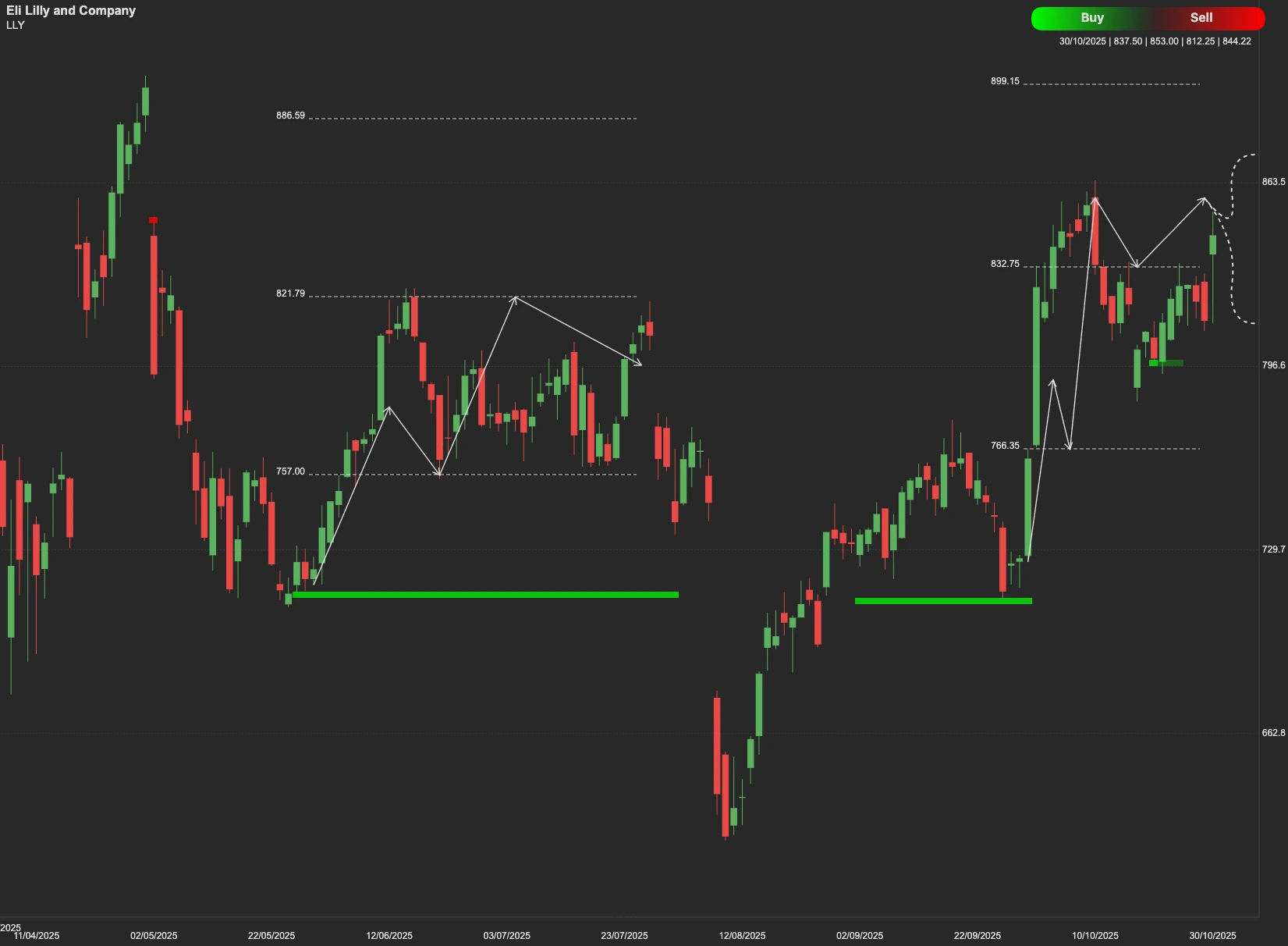
Technical Outlook
The current technical analysis identifies a key resistance level at $875. If the market translates the strong fundamental news into sustained buying pressure and the stock manages to decisively close and hold above the $875 resistance, it signals a continuation of the upward trend. In this scenario, the next target for LLY stock would be $920.
On the other hand, if the stock fails to push past the $875 resistance, it may seek support. The immediate support level to watch is $798. A deeper retracement could see the stock test the further support level at $781.
